Guillian-Barré Syndrome Management
M3 India Newsdesk Sep 11, 2023
This article provides an overview of Guillain-Barré syndrome (GBS), covering its clinical diagnosis, management, and subtypes, with a focus on its pathogenesis, clinical presentation, diagnostic methods, and treatment options.
Overview
Guillain-Barré syndrome (GBS) refers to a group of disorders affecting the peripheral nerves, with acute onset and a monophasic course, and is mostly demyelination, post-infectious in nature. The annual incidence of GBS is about 1.55/100,000 person-years and is higher in the younger population. Men are affected approximately 1.5 times more than women.
AIDP is the most common form of GBS. The other variants of GBS are:
The axonal variants
- Acute motor axonal neuropathy (AMAN)
- Acute motor-sensory axonal neuropathy (AMSAN)
The anti-GQ1b syndromes
- Miller-Fisher Syndrome
- Bickerstaff Encephalitis
- Pharyngeal-cervical-brachial weakness
Others
- Polyneuritis cranialis
- Pure sensory GBS
- Acute pandysautonomia
Pathogenesis
The neuropathy of GBS is commonly triggered after a preceding infection where there occurs an immune reaction to a shared epitope on the peripheral nerve (molecular mimicry).
- C.jejuni-related gastroenteritis is the most common event that triggers GBS. It is known to generate antibodies to specific gangliosides (GM1, GD1a, GalNac-GD1a, GQ1b, GD1b) which lead to the development of GBS and its subtypes.
- Other agents related to GBS include Haemophilus influenzae, Mycoplasma pneumoniae, and cytomegalovirus. Some cases have been reported after vaccination; however, the chances are significantly lower compared to GBS triggered by infections.
- In the demyelinating variants (AIDP, MFS), demyelination begins at the roots where the blood-nerve barrier is deficient. Demyelination is patchy but widespread. It leads to a blockade of saltatory conduction, leading to progressive conduction block and muscle weakness.
- The axonal variants (AMAN, AMSAN) are characterised by direct axolemmal injury due to the immune response. The primary targets are the nodes of Ranvier which leads to paranodal myelin detachment, node lengthening and sodium channel dysfunction which progresses to axonal degeneration.
Clinical presentation
1. GBS has a wide range of manifestations owing to various extents of injury to the neurons. AIDP typically presents with progressive symmetric (proximal & distal) weakness of limbs which is ascending in nature.
Symptoms associated with GBS
- Sensory symptoms like backache and radicular pain may be present. They are associated with reduced or absent deep tendon reflexes.
- Respiratory muscle involvement in severe cases may require mechanical ventilation.
- Cranial nerve involvement and autonomic dysfunction may be seen in some cases; sphincter involvement is rare.
- The AMAN variant of GBS presents with weakness predominantly in the legs.
- Sensory and autonomic involvement is minimal, however, reduced deep tendon reflexes are noted.
- In the AMSAN variant, there is much more severe sensory and motor nerve affection with autonomic involvement and a poor prognosis.
- The Miller-Fisher syndrome is defined by a triad of ataxia, areflexia and ophthalmoplegia.
- Facial and bulbar weakness can also be present. When these features are accompanied by altered sensorium, it is termed Bickerstaff Encephalitis. There can be overlapping features with AIDP as well.
- Other rare variants of GBS include the pharyngeal-cervical-brachial variant, acute autonomic neuropathy, and pure sensory neuropathy.
2. The natural history of GBS includes a monophasic course, antecedent diarrheal or respiratory illness and attainment of disease nadir by 28 days. Recovery may be prolonged, and may even take 6-12 months.
3. Various scoring systems are available to document the severity and prognosis of patients with GBS.
4. The GBS disability scale (Hughes et al.) provides an objective grading of deficits and severity of the illness.
5. The Erasmus GBS Respiratory Insufficiency Score (EGRIS) helps triage patients during the first week of disease and predict the probability of mechanical ventilation requirement.
6. The Modified Erasmus GBS Outcome Score (mEGOS) helps decide the long-term functional outcome of GB patients.
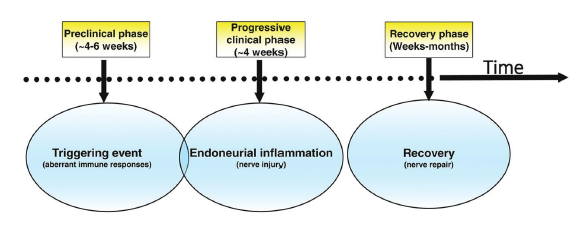
Investigations and diagnosis
- Diagnostic tests may not return positive results during the first week of illness, hence clinical judgement is of utmost importance of timely initiating immunotherapy.
- Nerve Conduction Studies with EMG help to confirm the involvement of peripheral nerves or roots, differentiate between the demyelinating and axonal subtypes and prognosticate regarding the extent of axonal injury.
- CSF studies typically show albuminocytological dissociation, i.e., an elevated protein with normal cell counts. CSF cell counts > 50 cells/mm3 should raise a suspicion of HIV, Lyme Disease, CMV or a leptomeningeal inflammatory process.
- Lastly, antiganglioside antibody measurements can provide supportive evidence in certain GBS subtypes, such as Miller-Fisher Syndrome (Anti-GQ1b) and AMAN (IgG anti-GM1 and anti-GD1a), although pending antibody results should not delay initiation of therapy in suspected cases.
Differential diagnosis
Although the onset and clinical manifestations of GBS usually help in making a straightforward diagnosis, one must always keep in mind the following differentials for GBS:
- Post-infectious Anterior Horn Cell diseases (West Nile virus, enterovirus, polio, rabies)
- Neuromuscular Junction disorders (Botulism, Myasthenia Gravis)
- Acute myopathies (Immune-mediated, infectious, myositis)
- Periodic Paralyses
- Acute onset Polyradiculopathies and Neuropathies (Critical Illness Neuropathy, Vasculitis, HIV, Lyme Disease, Toxic Neuropathies)
Treatment
- All patients with GBS require hospitalisation where ICU and Ventilator facilities are readily available. They require monitoring for BP, heart rate, respiratory functions, bulbar function, and progression of muscle weakness.
- Elective intubation may be provided for those at risk using the EGRIS score as mentioned above.
- Plasma Exchange and IVIg are the two recommended therapies for GBS, with both having equal efficacy as seen in head-to-head trials. IVIg is easier to administer and should be initiated within 2 weeks of disease onset (total dose of 2g/kg divided over 5 days).
- For patients presenting beyond 2 weeks of onset, Plasma Exchange is preferable (up to 4 weeks on onset).
- A standard protocol is to exchange 200 mL/kg to 250 mL/kg in four to five sessions over 7 to 14 days.
- Combination therapy of IVIg and Plasma Exchange should not be encouraged as they have not shown superior efficacy over individual therapies.
Prognosis and outcome
- Functional recovery from GBS is prolonged and takes weeks to months. Although more than 80% of patients recover completely, residual deficits can be seen in around 10% of patients. Another 10% may have a prolonged ICU stay with mechanical ventilatory support and a delayed recovery.
- Old age, rapidly progressive weakness, the need for mechanical ventilation, cardiovascular comorbidities, and infections increase mortality and have a poor prognosis. Axonal variants of GBS (AMAN, AMSAN) have lesser chances of recovery.
- Treatment-related fluctuations (i.e., a fall in the GBS disability scale after initial improvement) may be seen in some cases. Because of a lack of evidence, retreatment with the original modality is the usual treatment approach in these cases.
Disclaimer- The views and opinions expressed in this article are those of the author and do not necessarily reflect the official policy or position of M3 India.
About the author of this article: Dr. Annesh Bhattacharjee is a Consultant Neurologist from Guwahati.
-
Exclusive Write-ups & Webinars by KOLs
-
Daily Quiz by specialty
-
Paid Market Research Surveys
-
Case discussions, News & Journals' summaries