Choosing the right type of OCP: PCOS Society of India Consensus Statement
M3 India Newsdesk Jun 05, 2019
Taking into account the PCOS-metabolic syndrome association, Indian phenotype, and the risks of using OCPs in the presence of other confounding factors or comorbidities, the PCOS Society of India has published a statement intended to guide doctors in prescribing contraceptives to patients.
Oral contraceptive pills (OCPs) have been the first‑line therapy for concurrent treatment of menstrual irregularity, acne, and hirsutism in women with polycystic ovarian syndrome (PCOS), thus playing an important role in the symptom management of the PCOS women.
The association of PCOS with metabolic syndrome brings up the debate regarding the risks versus benefits of OCP use in PCOS women. Moreover, the presence of confounding factors such as obesity, smoking, diabetes, and other cardiometabolic comorbidities is a significant limitation in comparing the different OCPs as well as the risks and benefits associated with their use.
Acknowledging these aspects and considering the phenotype of Indian women, who tend to have a higher risk of metabolic syndrome and insulin resistance, the PCOS Society of India provides the guidance/recommendations for Indian practitioners regarding the use of OCP in women with PCOS. The guidance document has been published in the Journal of Human Reproductive Sciences.
What does the guidance document entail?
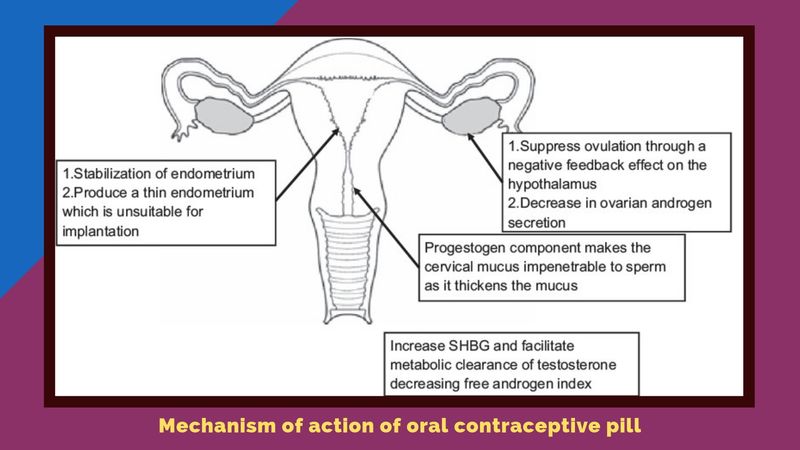
For several years, OCPs have played an important role in the symptom management of PCOS women. This is due to the fact that OCPs decrease the luteinizing hormone, reduce androgen production, and increase sex hormone‑binding globulin, which binds androgens.
Several new formulations of OCPs have been developed to decrease the side effects. This includes use of less androgenic progestins and lower doses of ethinyl estradiol.
These consensus recommendations help the health provider to choose the right type of OCPs, which will alleviate the symptoms with least side effects. It also gives insight into the indications, contraindications, and concerns regarding its short, intermediate and long-term use.
Pharmacology of OC pills
- There are two types of OCPs:
- Combined pills (combined oral contraceptive [COCP]), containing an estrogen and a progestogen
- Progestogen only pills
- The COCP is a combination of 2 types of steroids; ethinyl estradiol (EE) as the main estrogenic component, with the dose varying from 20 to 50 µg per tablet (usually 30–35 micrograms), and progestins essentially derivatives of 19‑nortestosterone (norethisterone, levonorgestrel, and more recently desogestrel, gestodene, norgestimate, and drospirenone)
Indications for the use of OCPs

Contraindications for the use of OCPs

Key recommendations
- Women aged 40 years and older can use COCPs but should follow up on a regular basis for CV risk, dyslipidaemia, and thromboembolism
- COCP use may decrease bone mineral density (BMD) in adolescents, especially in those choosing very low‑dose formulations
Drug interactions
Pharmacological interactions between OCPs and other compounds may be of two types:
- Drugs may impair the effectiveness of the OCPs, or
- OCPs may interfere with the metabolism of other compounds
Interactions of the first kind are due to interference with the absorption, metabolism, or excretion of estrogen, and interactions of the second kind are due to competition for metabolic pathway.
- For women on antiepileptic drugs (AEDs) that may compromise the efficacy of OCPs, high‑dose pills containing at least 50 mcg of EE should be prescribed.
- Dose of lamotrigine may be doubled to maintain steady therapeutic levels, but its side effects can occur during pill‑free interval, and the patient should remember to reduce the lamotrigine dosage during these days.
- OCPs users may require larger doses of acetaminophen, aspirin temazepam, paracetamol, morphine, and clofibric acid.
- Women on protease inhibitors should be advised to use the alternative methods of contraception.
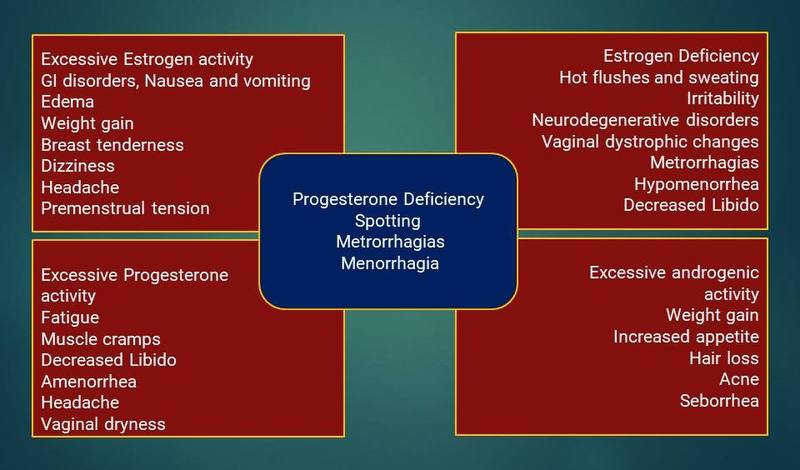
Side effects
Side effects of oral contraceptives vary depending on the dose and type of hormone used in the OCPs. Side effects, in general, include breast tenderness, bloating, nausea, weight gain, migraine, insulin resistance, increased blood sugars, dyslipidaemia, VTE, cardiovascular disease (CVD), vaginal spotting, and abnormal bleeding. Mild increase in blood pressure might occur with some newer OCPs formulations.
The key recommendations are:
- Global CVD risk needs to be considered especially in women who are
- at risk of CVD
- older age
- presence of diabetes, hypertension, and dyslipidaemias
- those who are obese
- those who smoke before commencing the OCP
- In smokers, the third‑generation OCPs may have deleterious effects on coagulation and should be avoided
- Specific types or dose of progestins, estrogens, or combinations of COCP cannot currently be recommended with inadequate evidence in adults and adolescents with PCOS, and the practice should follow general population guidelines
General Counseling
To achieve maximum benefits and safety for each individual patient, for non-contraceptive use of COCP, detailed counseling regarding risks, benefits, and contraindications should be done.
- Counseling regarding risks, benefits, and contraindications should be done in detail before starting OCPs
- Important to discuss the interactions of OCPs with other drugs
Investigations before initiation
- Before prescribing a COCP (Combined OCP), obtain a thorough past medical/family history, calculate BMI and blood pressure
- Pelvic examinations and Pap smears are not required before prescribing COCP medications
- Measuring blood sugar levels and OGTs may not be recommended unless the women are overweight or have other diabetes risk factors
Starting and stopping of therapy with oral contraceptive pills
- OCPs are ideally started on any day between day 1 and 5 of the cycle, given for 21 days and stopped. This will be followed by withdrawal bleed.
- A new packet of pills is started counting the 1st day of withdrawal bleed as day 1.
- The patient should be called for follow‑up after 3 months for the assessment of blood pressure and enquired for the presence of any other problems.
- Thereafter, annual visit is required for assessing side effects, checking blood pressure, and evaluating glucose intolerance and lipids.
The key recommendations include:
- Proper patient selection is necessary to minimise risks associated with COCP use
- In adolescents, it is best to wait for 2 years after menarche to prevent reduction in peak bone mass
- It is necessary to make women aware of increased risk of cardiometabolic disorders when on OCPs
- OCPs need to be stopped in the presence of any side effects or if abnormal glucose tolerance or dyslipidemia is detected
Long‑term concerns
Three issues that need to be addressed include:
- Cardiovascular risk
- Thrombosis with risk of embolism
- Cancer risks
Other recommendations are:
- Smoking, hypertension, and diabetes are risk factors for CV risk. Controlling these factors reduces the long‑term risk of MI and stroke.
- Obese women who use COCP are more likely to experience VTE than obese nonusers. COCP should be used with caution in obese PCOS.
- Mild dyslipidaemia is not a contraindication to use OCP, but the women should be made aware of the fact that OCPs can change lipid profile and therefore should be monitored based on metabolic risk profiles.
- Higher risk of cervical cancer reported in women who use COCP, especially with an increased duration of COCP use. Therefore, restrict the use of COCP to shorter duration.
- Health‑care professionals should be aware of impacts of the OCP on CVD, BMD (in adolescents), venous thrombosis risk, hypertension, lipid profile, and endometrial, breast (during or within 10 years of use), and liver cancer.
Follow‑up on oral contraceptive pill therapy
No evidence exists whether a routine follow‑up after initiating COCP is essential. It is best to schedule a visit for the assessment of blood pressure and any other problems after 3 months or earlier if required.
- Specific populations who might benefit from more frequent follow‑up visits include adolescents and those with certain medical conditions
- Monitoring blood pressure and BMI is important for women using COCPs
Choice of oral contraceptive pill for non-contraceptive Indications
Acne
Estrogen‑containing COCP are effective and recommended in the treatment of inflammatory acne
Both EE/DRSP (drospirenone) and EE/cyproterone acetate combinations improve hyperandrogenic parameters significantly without affecting the insulin resistance adversely in Indian women
Hirsutism
- Epilatory measures need to be used in conjunction with OCPs
- All OCPs appear to be equally effective for hirsutism, and the risk of side effects is low
- Progestins preferred are the antiandrogen progestins such as cyproterone acetate and drospirenone
- Preferable, to use an OC containing a progestin with low‑androgenic activity (e.g., norethindrone acetate, ethynodiol diacetate, desogestrel, gestodene, norgestimate)
- If hirsutism does not respond to COCPs despite 6 months of monotherapy with an OCP, add antiandrogens
Menstrual irregularity and menorrhagia
- COCP is effective in treating irregular cycles and menorrhagia
- OCPs with the first and second‑generation progestins are preferred in acute menorrhagia and a few subsequent cycles. Once bleeding is relatively controlled, one can use the third and fourth generation OCP
- In adolescents and women with menorrhagia, it is important to rule out haemoglobinopathies and coagulopathies
- Levonorgestrel intrauterine device can be offered in those with menorrhagia
Oral contraceptive pills for long‑term therapy in polycystic ovarian syndrome
The two main concerns when COCPs are used in PCOS women are decrease in bone density and fear of VTE (Venous thromboembolism).
Moreover, OCPs use is also linked to cervical, breast, and liver cancers. Long‑term use of OCPs has also been associated with increased risk of glaucoma, especially when used for 3 years or more.
- It is essential to carefully monitor PCOS women on OCPs for BMD, venous thrombosis, hypertension, lipid profile, and breast, cervical, and liver cancer
- Routine ophthalmic examination (at least yearly) for monitoring glaucoma
Infertility management
During treatment with hormonal contraceptives, serum concentrations of follicle‑stimulating hormone (FSH), LH, and estradiol decrease. Thus, COCP is associated with suppression of ovarian and endometrial function.

- Ovarian reserve markers are lower in women using sex steroids for contraception
- OCPs pre-treatment for 2 months followed by clomiphene citrate (CC) at dosage of 100 mg/day on days 5‑9 of the cycle is associated with excellent ovulation rates and pregnancy rates
- OCPs cyclical pre-treatment before ART could improve pregnancy outcome due to reduction of LH, hyperandrogenism, and antral follicle excess
- COCP pre-treatment in antagonist protocols was associated with a lower rate of LBR or ongoing pregnancy than no pre-treatment and this is related to the pill‑free interval
- Benefits of synchronisation of follicular cohort and cycle scheduling must be weighed against the drawbacks with pre-treatment OCP in GnRH antagonist
- COCP in FET ((Frozen Embryo Transfer) cycles may reduce the risk of pregnancy loss
- COCP reduce endometriosis‑associated pain, dyspareunia, and dysmenorrhea
Use of oral contraceptive pills in lean and obese women
The prevalence of obesity is higher in PCOS women with increased visceral adiposity. OCPs when used:
- desire increased awareness of the patients to the side effects and complications, in both lean and obese women
- is associated with no change in body fat distribution or decrease in visceral fat
- has additional metabolic risk and should be carefully monitored by appropriate surveillance including blood pressure and weight
Oral contraceptive pills for added benefit of contraception
OCPs, apart from the other benefits also, offer contraception to those PCOS women who do not want to conceive.
- COCP offer contraception to those PCOS women who do not want to conceive
- Progestin‑only contraceptive pill or levonorgestrel‑containing intrauterine system can be used in PCOS women where the use of estrogen is contraindicated or when androgen excess does not exist
- Depot medroxyprogesterone and etonogestrel‑containing implant should not be used in women with PCOS
Prevention of cancer in Polycystic Ovarian Syndrome
Most PCOS women have anovulatory cycles with disruption of normal reproductive physiology, which increases the risk of the development of cancer of the endometrium, ovary, and/or breast, either directly or mediated by its associated reproductive metabolic alterations. OCPs have protective beneficial effect against endometrial and ovarian cancer.
Use of other drugs with oral contraceptive pills in Polycystic Ovarian Syndrome
- Glucocorticoids and GnRH analogs should not be used regularly with OCPs for the treatment of hyperandrogenaemia.
- Use of glucocorticoids should be restricted only to women who have NCCAH (nonclassical congenital adrenal hyperplasia).
- Antiandrogens are recommended along with COCP in women presenting with moderate or severe hirsutism, women presenting with a milder hirsutism, who do not reach a satisfactory control of hair growth using OCPs alone after 6‑9 months of treatment.
- Metformin or other insulin sensitisers should not be used as therapy for hyperandrogenaemia as the evidence is unconvincing and possibly not superior to placebo.
- Metformin can be used with COCP in PCOS women for management of cardiometabolic factor and prevention of weight gain and in those PCOS women who are obese and overweight. Use of metformin may be relevant in Asian Indians due to high risk.
Practice points to note
- In PCOS women who do not desire fertility, OCPs are the first‑line treatment to improve clinical symptoms of androgen excess such as acne and hirsutism and also help in regulating the menstrual cycles and protecting the endometrium against effects of unopposed estrogen action.
- Estrogen content in the OCP increases SHBG levels, thus decreasing the free circulating androgens. The progestin component of OCP suppresses the secretion of LH and decreases ovarian androgen production.
- Menstrual irregularity gets corrected immediately, but acne and hirsutism may take a minimum of 6 months for its effects to be seen.
- Low‑dose COCP containing neutral or antiandrogenic progestins may be the OCPs of choice in the treatment of PCOS.
- Despite the potential adverse cardiovascular and metabolic effects of OCPs, the current evidence suggests that the benefits outweigh the risks for its use in most PCOS women.
- Use of OCPs in PCOS women should be individualised after risk stratification and not used if any contraindications exist.
-
Exclusive Write-ups & Webinars by KOLs
-
Daily Quiz by specialty
-
Paid Market Research Surveys
-
Case discussions, News & Journals' summaries